Science Class X
How do nerve impulses travel in the body
How do nerve impulses travel in the body ? Explain.
Read more …Why is the use of iodised salt advisable
Why is the use of iodised salt advisable ? Name the disease caused due to deficiency of iodine in our diet and state its one symptom.
Read more …What is hydrotropism
What is hydrotropism ? Design an experiment to demonstrate this phenomenon.
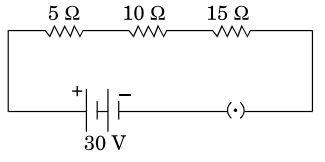
Read more …Consider the given circuit and find the current flowing
Consider the given circuit and find the current flowing in the circuit and potential difference across the 15 Ω resistor when the circuit is closed.
Calculate the equivalent resistance of the following network
Calculate the equivalent resistance of the following network :

A teacher provided acetic acid, water, lemon juice, aqueous solution
A teacher provided acetic acid, water, lemon juice, aqueous solution of sodium hydrogen carbonate and sodium hydroxide to students in the school laboratory to determine the pH values of these substances using pH papers. One of the students reported the pH values of the given substances as 3, 12, 4, 8 and 14 respectively. Which one of these values is not correct ? Write its correct value stating the reason.
Read more …Placing duly cleaned strips of aluminium, copper, iron and zinc
What would a student report nearly after 30 minutes of placing duly cleaned strips of aluminium, copper, iron and zinc in freshly prepared iron sulphate solution taken in four beakers ?
Read more …What is observed when a pinch of sodium hydrogen carbonate
What is observed when a pinch of sodium hydrogen carbonate is added to 2 mL of acetic acid taken in a test tube ? Write chemical equation for the reaction involved in this case.
Read more …After examining a prepared slide under the high power
After examining a prepared slide under the high power of a compound microscope, a student concludes that the given slide shows the various stages of binary fission in a unicellular organism. Write two observations on the basis of which such a conclusion may be drawn.
Read more …List four steps of obtaining germinating dicot seeds
List in proper sequence four steps of obtaining germinating dicot seeds.
Read more …List four precautions while preparing a temporary mount of a leaf peel
List four precautions which a student should observe while preparing a temporary mount of a leaf peel to show stomata in his school laboratory.
Read more …The current flowing through a resistor connected in a circuit
The current flowing through a resistor connected in a circuit and the potential difference developed across its ends are as shown in the diagram by milliammeter and voltmeter readings respectively:
- What are the least counts of these meters ?
- What is the resistance of the resistor ?

Carbon has four electrons in its valence shell
Carbon has four electrons in its valence shell. Which type of compounds can be formed by carbon atom and why ? Give any one example of such compounds.
Read more …An object is kept 40 cm in front of a concave mirror of focal length 20 cm
An object is kept 40 cm in front of a concave mirror of focal length 20 cm. Find the position and nature of the image. Draw a ray diagram to show the formation of image in this case.
Read more …In the reaction: MnO2 + 4HCl → MnCl2 + 2H2O + Cl2
In the reaction:
MnO2 + 4HCl → MnCl2 + 2H2O + Cl2
- Name the compound (i) oxidised, (ii) reduced.
- Define oxidation and reduction on its basis.
For the preparation of cakes, baking powder is used
(a) For the preparation of cakes, baking powder is used. If at home your mother uses baking soda instead of baking powder, how will it affect the taste of the cake and why?
(b) How is baking soda be converted into baking powder?
(c) What makes the cake soft and spongy?
Read more …In the electrolysis of water
In the electrolysis of water,
- Name the gas collected at anode and cathode?
- Why is the volume of gas collected at one electrode double than the other?
- What would happen if dil H2SO4 is not added to water?
pH has a great importance in our daily life
"pH has a great importance in our daily life". Explain by giving three examples.
Read more …A compound which is prepared from gypsum has the property of hardening
A compound which is prepared from gypsum has the property of hardening when mixed with a proper quantity of water. Identify the compound and write its chemical formula. Write the chemical equation for its preparation. Mention any one use of the compound.
Read more …1 g of solid sodium chloride is taken in a clean and dry test tube
1 g of solid sodium chloride is taken in a clean and dry test tube and 2 mL of conc. sulphuric acid is added to it. If the gas evolved is tested first with dry and then with wet blue litmus paper, in which case will the litmus paper change colour? Give reason for your answer. What inference can be drawn about the nature of the evolved gas ? Support your answer with chemical equation for the reaction.
Read more …What happens when hydrogen is added to a vegetable oil in the presence of nickel
What happens when hydrogen is added to a vegetable oil in the presence of nickel? Name the reaction and write one difference between the physical property of the vegetable oil and the product obtained in this reaction. Write the role of nickel in this reaction.
Read more …An object of height 1.2 m is placed before a concave mirror
An object of height 1.2 m is placed before a concave mirror of focal length 20 cm so that a real image is formed at a distance of 60 cm from it. Find the position of an object. What will be the height of the image formed?
Read more …Rohit focused the image of a candle flame on a white screen
Rohit focused the image of a candle flame on a white screen using a convex lens. He noted down the position of the candle, screen and lens as under:
Position of candle = 26.0 cm
Position of convex lens = 50.0 cm
Position of screen = 74.0 cm
i) What is the focal length of the convex lens?
ii) Where will the image be formed if he shifts the candle towards the lens at a position of 38 cm?
iii) Draw a ray diagram to show the formation of the image in case (ii) as said above?
How Did Mendel Explain That Trait Is Inherited But Not Expressed
How did Mendel explain that it is possible that a trait is inherited but not expressed in an organism?
Read more …Distinguish Between Acquired Traits And Inherited Traits
With the help of one example for each, distinguish between the acquired traits and the inherited traits. Why are the traits / experiences acquired during the entire lifetime of an individual not inherited in the next generation? Explain the reason of this fact with an example.
Read more …What are acquired traits? Why are these traits generally not inherited over generations
What are acquired traits? Why are these traits generally not inherited over generations? Explain.
Read more …We wish to obtain an equal sized inverted image of a candle flame on a screen
We wish to obtain an equal sized inverted image of a candle flame on a screen kept at distance of 4 m from the candle flame.
- Name the type of lens that should be used.
- What should be the focal length of the lens and at what distance from the candle flame the lens be placed.
- Draw a labelled diagram to show the image formation in this case.
A 5 cm tall object is placed at a distance of 30 cm from a convex mirror
A 5 cm tall object is placed at a distance of 30 cm from a convex mirror of focal length 15 cm. Find the position, size and nature of the image formed.
Read more …What is electrical resistivity? Derive its SI unit
What is electrical resistivity? Derive its SI unit. In a series electrical circuit comprising a resistor made up of a metallic wire, the ammeter reads 100 mA. If the length of the wire is doubled, how will the current in the circuit change? Justify your answer.
Read more …What would be the readings of the ammeter and the voltmeter
Consider the following circuit:

What would be the readings of the ammeter and the voltmeter when key is closed? Give reason to justify your answers.
Read more …Calculate the total cost of running the following electrical devices
Calculate the total cost of running the following electrical devices in the month of September, if the rate of 1 unit of electricity is Rs 6.00.
- Electric heater of 1000 W for 5 hours daily.
- Electric refrigerator of 400 W for 10 hours daily.
Improvements in our lifestyle have resulted in greater amounts of waste generation
"Improvements in our lifestyle have resulted in greater amounts of waste generation." Give two examples to support the given statement. Suggest one change that we can incorporate in our lifestyle in order to reduce non biodegradable waste.
Read more …What do you understand by Watershed Management
What do you understand by "Watershed Management"? List any two advantages of watershed management.
Read more …What is “Sustainable Management of Natural Resources”
What is “Sustainable Management of Natural Resources”? Why is it necessary? Which one out of reuse and recycle, would you practise in your daily life and why?
Read more …State ways to prevent the rusting of iron
State two ways to prevent the rusting of iron.
Read more …What is reactivity series
What is reactivity series? How does the reactivity series of metals help in predicting the relative activities of various metals?
Read more …Suggest different chemical processes used for obtaining a metal from its oxides
Suggest different chemical processes used for obtaining a metal from its oxides for metals in the middle of the reactivity series and metals towards the top of the reactivity series. Support your answer with one example each.
Read more …List in tabular form chemical properties on the basis of which
List in tabular form any three chemical properties on the basis of which metals and non-metals are differentiated.
Read more …What was the basis of Mandelẽev’s classification of elements
(a) What was the basis of Mandelẽev’s classification of elements?
(b) List two achievements of Mandelẽev’s periodic tables.
(c) List any two observations which posed a challenge to Mandelẽev’s periodic law.
Read more …Write Number Of Periods And Groups In Modern Periodic Table
Write the number of periods and groups in the Modern Periodic Table. How does the metallic character of elements vary on moving (i) from left to right in a period, and (ii) down a group? Give reason to justify your answer.
Read more …Na, Mg and Al Are Elements Of 3rd Period
Na, Mg and Al are the elements of the 3rd period of the Modern Periodic Table having group number 1, 2 and 13 respectively. Which one of these elements has the (a) highest valency, (b) largest atomic radius, and (c) maximum chemical reactivity? Justify your answer stating the reason for each.
Read more …Differentiate between the arrangement of elements in Mendeleev’s periodic table
Differentiate between the arrangement of elements in Mendeleev’s periodic table and Modern periodic table.
Read more …Explain giving justification the trends in the following properties of elements
Explain giving justification the trends in the following properties of elements, on moving from left to right in a period, in the Modern periodic Table.
- Variation of valency.
- Change of atomic radius.
- Metallic to non-metallic character.
- Electronegative character.
- Nature of oxides.
Write the mechanism by which fishes breath in water
Write the mechanism by which fishes breath in water.
Read more …Name the balloon likes structures present in lungs
Name the balloon likes structures present in lungs. List its two functions.
Read more …Name the respiratory pigment and write its role in human beings
Name the respiratory pigment and write its role in human beings.
Read more …Explain the ways in which glucose is broken down in absence of oxygen
Explain the ways in which glucose is broken down in absence of oxygen.
Read more …List differences between arteries and veins
List three differences between arteries and veins.
Read more …Name the process and explain the type of nutrition found in green plants
(a) Name the process and explain the type of nutrition found in green plants. List the raw materials required for this process. Give chemical equation for the mentioned process.
(b) Write three events that occur during this process.
Read more …Page 2 of 4